Lifespan is determined by the state of the cellular energy system
Over time, the efficiency of mitochondria – the ‘energy stations’ of cells – decreases, which is associated with the aging process and the development of age-related diseases. Scientists from Harvard University (USA) investigated what happens to these essential cellular structures when calorie restriction is used as a recognised way to increase longevity.
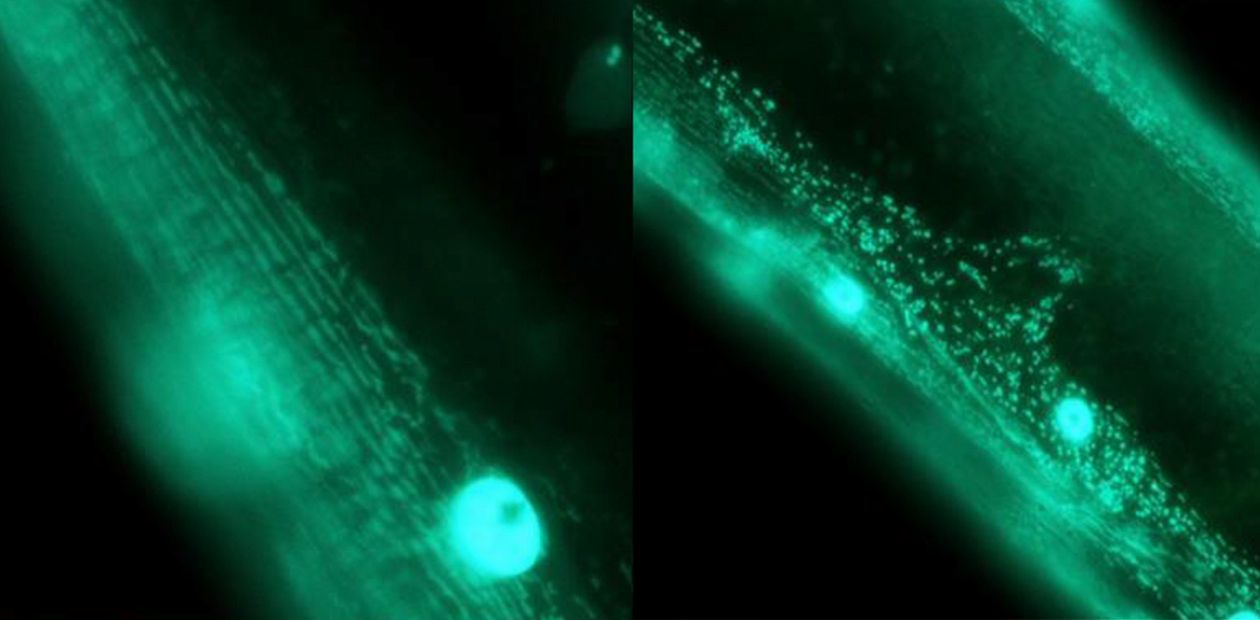
Muscle tissue of C. elegans nematodes. Left, normal mitochondria integrated into networks; right, fragmented mitochondria in a pathologically altered organ. Staining with green fluorescent protein
Mitochondria are known to produce ATP molecules, a universal source of energy for all biochemical processes in the body, including developmental processes, metabolic regulation and cell division. For a long time mitochondria were represented as discrete organelles, but recently the concept of mitochondrial dynamics, according to which mitochondria can fuse or, on the contrary, divide into parts depending on the energy needs of the cell, has gained recognition.
It turned out that in most cells mitochondria are organised into a three-dimensional, dynamically changing network, the parameters of which are under the control of AMPK (AMP-activated protein kinase), a key regulatory protein of energy balance. Disturbances in mitochondrial dynamics, the ‘wrong appearance’ of organelles, and their excessive fragmentation are signs of aging and are thought to contribute to the development of age-related pathologies, including Alzheimer’s and Parkinson’s disease.
It is also known that the aforementioned AMPK protein plays a significant role in prolonging life by restricting the caloric content of the diet. This protein can be activated not only by a low-calorie diet, but also directly, for example, with metformin, a type 2 diabetes medication. In the latter case, it has been shown that the administration of this drug does lead to an increase in the life expectancy of laboratory mice.
A particularly convenient model for studying ageing is the free-living nematode worm Caenorhabditis elegans, whose normal life span does not exceed two weeks. It is on these small roundworms scientists have studied the relationship between life span and the nature of changes in mitochondrial networks.
It turned out that the activation of AMPK really contributed to the increase in animal longevity by maintaining a stable dynamic balance in mitochondrial networks, which was accompanied by an increase in fatty acid oxidation. It is about the balance of mitochondrial rearrangement processes, because even if we artificially maintain mitochondria in the state of fusion, more typical for healthy, ‘young’ cells, the effect in life extension cannot be achieved. And here AMPK activation does not help either by dietary restriction or by genetic-engineering manipulations.
The results have allowed a better understanding of what happens in cells during dietary restriction, and opened new opportunities for finding therapeutic strategies to reduce the risk of age-related pathologies. In the nearest plans of scientists – to lay their research, but already on mammals. Because, whatever you say, but your shirt is always closer to your body.
Source: Science and Life
Don’t miss the most important science and health updates!
Subscribe to our newsletter and get the most important news straight to your inbox
Published
July, 2024
Duration of reading
About 2-3 minutes
Category
Aging and youth
Share

